The White House’s fiscal year 2027 budget proposal, released earlier this month, would cut the National Institutes of Health (NIH) funding by ~$5 billion. This is a meaningful cut, though far smaller than the roughly $18 billion reduction proposed for 2026, which Congress ultimately rejected in favor of a modest increase. Whether Congress holds the line again remains uncertain.
This moment, however, invites a broader question: beyond the size of the budget, are there ways NIH can use its resources more effectively?
NIH should expand its use of “pull” funding, starting with generic drug repurposing, while continuing to support its core “push” funding model. With an annual budget of roughly $40 billion, NIH is the world’s largest funder of biomedical research. Its traditional grant-based approach, often focused on areas the private sector won’t invest in, has helped drive breakthroughs including the first HIV/AIDS drugs. Given its significant role in biomedical research, even modest improvements in how NIH allocates its funding could have outsized impact.
What is “push” and “pull” funding?
Most of NIH’s budget flows through push funding such as grants awarded upfront. This model has powered decades of discoveries but requires funders to decide in advance who is best positioned to solve a problem and what approach is most likely to work.
Pull funding takes a different approach. Instead of paying upfront, it pays based on outcomes. An agency using pull funding would define a goal, such as demonstrated adoption of a drug for a new use, and offer a reward to whoever achieves it.
Push and pull funding are complements, not substitutes. Push funding is most valuable when NIH can identify strong projects using publicly available information, or in early-stage science where both public and private knowledge are limited. Pull funding is most valuable when researchers have private information that NIH cannot easily access. Since it only pays for success, pull funding filters for the most promising ideas.
In other words, push funding supports the promising opportunities NIH can already see. Pull funding helps crowdsource outside information and effort.
A useful analogy comes from the Wild West, where sheriffs pursued the most promising leads themselves but also posted bounties to draw in information and effort from outsiders.
Why focus on drug repurposing?
Generic drug repurposing represents a clear market failure, in need of public funding, and is also a natural fit for pull funding.
Why does generic drug repurposing require public funding? When a drug’s patent expires, “generic” competitors enter the market and drive prices down. With low prices and no exclusive rights, companies cannot profit enough from a new use approval to justify funding expensive clinical trials. Promising treatments go unstudied not because the science is weak, but because the business case is missing.
Figure 1. The probability of approval for new use decreases after market exclusivity expiration
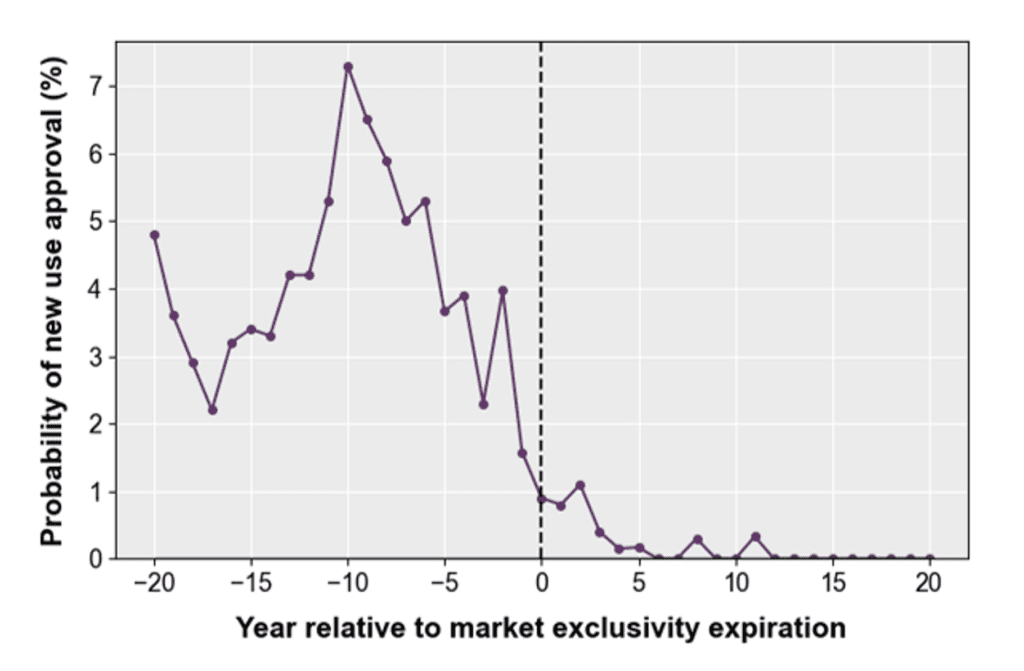
Source: Budish, et al. 2025
Note: A standard patent expires after 20 years.
Why is pull funding especially well-suited for drug repurposing? The key advantage of pull funding is that it taps dispersed, private knowledge. Clinicians may spot patterns long before they are formally tested. For example, decades after aspirin was introduced, Dr. Lawrence Craven observed its effects on bleeding and suggested it could prevent heart attacks. Years later, researchers confirmed this idea, and aspirin now reduces the risk of a second heart attack by about 20 percent for millions of Americans.
Additionally, companies may hold similar untapped insights. They have unpublished clinical trial data and analyze real-world data.[1] Both can provide insights into promising new uses.
Pull funding can also improve adoption. Even when research identifies new uses for generic drugs, clinicians are often slow to prescribe them. For example, metformin for prediabetes, a use identified through NIH-supported research, is used in only about 2 percent of eligible patients despite its significant benefits. Pull funding that rewards clinical impact, not just trial results, can help close this gap.
What a pilot could look like
A practical starting point is an NIH pilot that offers financial rewards for successful generic drug repurposing, with payments tied to impact and contingent on FDA approval. Eligibility should be broad, open to universities, startups, nonprofits, and established companies, since the strength of pull funding lies in attracting contributors the funder cannot identify in advance.
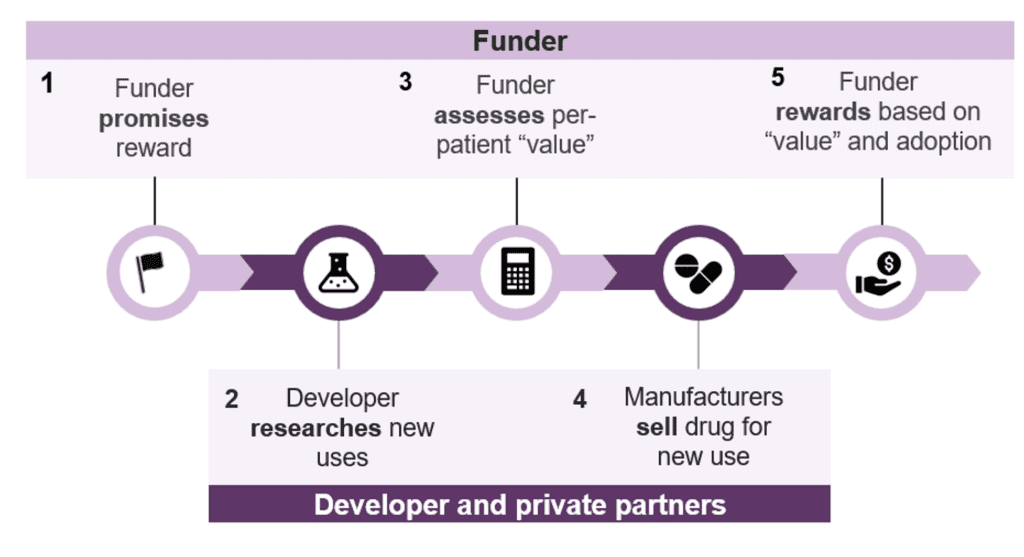
Pull funding could work as follows. First, the funder would commit to reward research sponsors based on the expected health impact of a repurposed drug. Second, in response, researchers would investigate new uses for generic drugs. Third, clinical trial data would then be used to assess impact. Fourth, if successful, manufacturers would bring the drug to market for the new use. Fifth and finally, NIH would reward the entity that generated the evidence (i.e., the trial sponsor), rather than the manufacturer, based on the drug’s measured value and real-world adoption.
Figure 2. Key design features of a pull funding model for generic drug repurposing
The generic drug repurposing program could further target areas with especially weak commercial incentives, such as drugs for healthcare emergencies and neglected tropical diseases. NIH already plays a central role in these disease areas through the National Institute of Allergy and Infectious Diseases.
This program may need new legislation to begin, but it’s possible that NIH could use existing authority, such as other transaction authority and/or prize authority, to run these programs. It could also coordinate with other US agencies, including the Centers for Medicare and Medicaid Services, Biomedical Advanced Research and Development Authority, and the Department of Defense. For example, as discussed in our white paper, Medicare and Medicaid’s Innovation Center could fund generic drug repurposing when it presents cost-savings. The NIH could then complement this effort by supporting high health-impact opportunities that do not yield immediate cost-savings.
Conclusion
At a time of budget pressure, it is important to consider how NIH can use its budget most effectively. Pull funding offers a practical complement to existing push funding, and drug repurposing is a natural place to start. By rewarding results rather than proposals, pull funding can surface hidden information, accelerate drug repurposing, and ensure proven treatments are actually used.
To learn more, please reach out to Sarrin Chethik: schethik@marketshapingaccelerator.org.
[1] While companies are required to publish clinical trial data, this only began in 2007 and, even since then, only about 45 percent of trials report results within one year.

